A new type of flu vaccine could save thousands of lives and reduce the number of GP consultations for flu by nearly 22,000 per year in the UK, according to new research.
The research found that the new vaccine was highly cost-effective at NICE thresholds and would reduce the number of deaths attributable to flu, as well as the number of GP consultations.
The new vaccine, Fluad, uses an adjuvant to help the body produce a stronger immune response against flu when administered. It is the only adjuvanted flu vaccine approved for use globally and will be available in the UK for use in the 2018/2019 flu season.
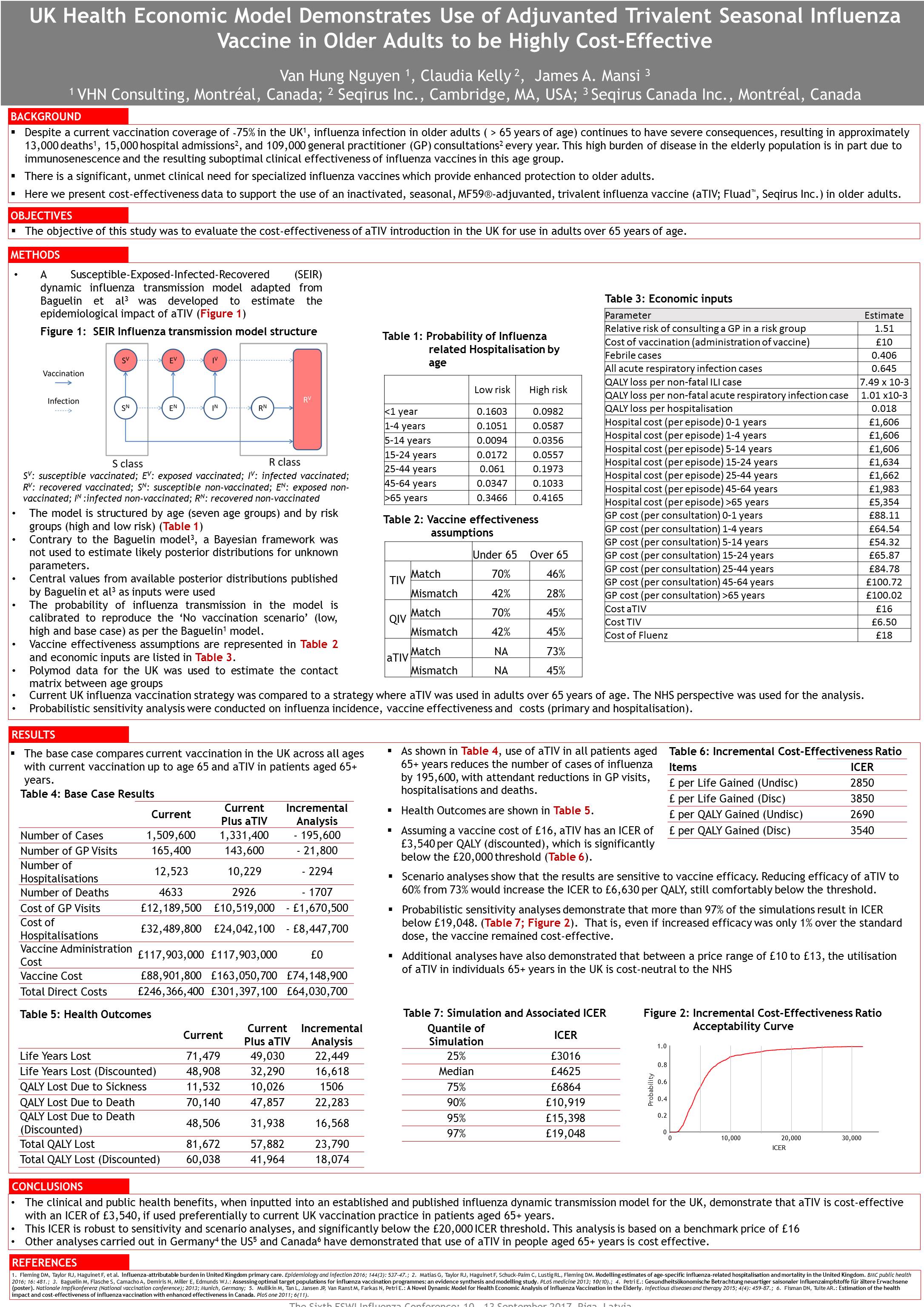
The research, presented this week at the European Scientific Working group on Influenza (ESWI) conference 2017, was based on health economic modelling. It found that 195,000 cases of flu could be avoided with the new vaccine, as well as 21,800 GP consultations and just over 1700 deaths. They also found that administering Fluad to over-65s would fall well below NICE’s cost-effectiveness threshold, costing £3540 per quality-adjusted life year gained.
Research poster presented at ESWI 2017 conference 12th September – click to view full size
In other modelling research also presented at the ESWI conference, the researchers predicted that the adjuvanted vaccine would induce greater antibody levels and therefore a greater level of clinical protection than an equivalent non-adjuvanted vaccine.
Dr Marco Barbieri, of the Centre for Health Economics at the University of York, commented: ‘Health economic analyses consistently show that more effective flu vaccines are very good value in older patients
‘They reduce the number of GP consultations, A&E visits and episodes of serious illness and, of course, they help older people to remain healthy and independent.
‘These data suggest that use of an adjuvanted influenza vaccine is a cost-effective option for the NHS immunisation programme.’
The Joint Committee on Vaccination and Immunisation acknowledged in their June meeting that cost-effectiveness data on adjuvanted vaccines would be useful in optimising the vaccination programme for over-65s and agreed to review the evidence at their October meeting.
The research comes as PHE announced this month that the flu vaccine was not significantly effective in over-65s last winter.