GPs told to switch to US nasal flu vaccine
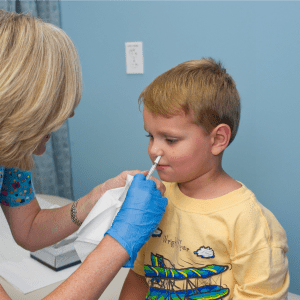
Practices are being forewarned that central supplies of Fluenz Tetra, the nasal flu vaccine for childhood flu immunisations, will soon run out and that they will need to order a US version of the vaccine instead.
Public health chiefs stressed the US vaccine – FluMist Quadrivalent – is exactly the same as Fluenz Tetra. However, they advised that GPs should ignore the expiry dates printed on packages of FluMist, as these will be inaccurate.
This is because the switchover has messed up the supply schedule, causing a ‘mismatch between the actual expiry date and that printed on the packaging and labelling’, Public Health England said in latest Vaccine Update.
Instead practices are advised simply not to use any FluMist Quadrivalent after 24 February 2016 – and that the vaccine supplier will start collecting unused stocks a month before this ‘to help ensure that no time-expired vaccine remains in circulation’.
GPs are also advised to quarantine any unused FluMist before 24 February ‘to avoid accidental administration prior to collection’.
In addition, GPs must refer to UK prescribing information – which will be included with FluMist packages – as this differs from the US information, and make sure to get an amended Patient Group Direction form signed off by NHS England as well as the authorising GP and prescriber.
However, the vaccine is administered in exactly the same way as Fluenz, while consent forms and recording of vaccinations remain the same.
The Fluenz supply problem is the latest in a series of shortages of the vaccine that has beset the childhood seasonal flu programme since its introduction in 2013.
Last year, GPs faced an immediate cap on how many vaccine packs they could order at the beginning of the flu season, despite PHE urging them to make sure as many children were vaccinated before Christmas because of the vaccine’s short shelf-life.
Visit Pulse Reference for details on 140 symptoms, including easily searchable symptoms and categories, offering you a free platform to check symptoms and receive potential diagnoses during consultations.









