‘My stomach hurts when I eat’ – managing chronic pancreatitis
Case
A 62-year-old female with a background of type 2 diabetes and COPD has returned again to her GP with grumbling abdominal pain plus occasional radiation to the back and some undefined weight loss over the last six months. She reports a loss of appetite as the pain is exacerbated by eating. Other than weight loss there are no alarms symptoms and a recent gastroscopy and abdominal ultrasound based on an earlier consultation were normal. She has been seen and discharged by the local gastroenterology service with a presumed diagnosis of IBS and counselled on self-management. Her bowel habit is unchanged, but she describes passing fluffy stools. Her only medications are metformin and occasional inhalers. The patient is a retired school cleaner, a lifelong smoker but has never drunk alcohol to excess. There is no family history of note.
On examination she is comfortable at rest and now has a normal BMI, although previously had been significantly overweight. There are no obvious palpable masses in the abdomen.
The question the GP faces is how to proceed in the management of this patient with insidious pain and weight loss but normal screening tests including upper endoscopy. Due to on-going pain and weight loss the decision to re-refer is made.
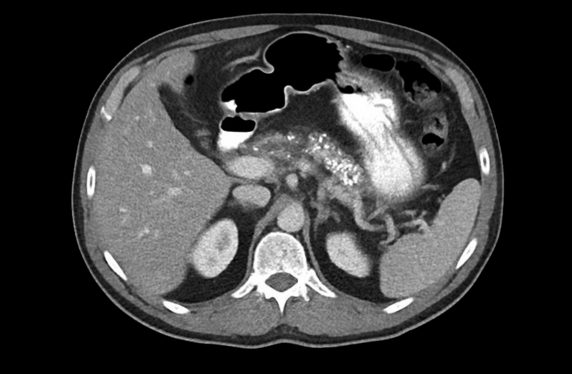
This time, a CT was arranged and showed diffuse pancreatic calcifications and atrophy. Further blood tests identified a low albumin and magnesium and the faecal elastase (FE) was 50 (normal >200 ug/g). Referral to the regional pancreatobiliary unit was made where the diagnosis of chronic pancreatitis was established. The patients’ health improved with regular analgesia, and taking pancreatic enzyme replacement (PERT), as well as a period of dietetic advice.
The problem
Chronic pancreatitis is a ‘disease of the pancreas in which recurrent inflammatory episodes result in replacement of the pancreatic parenchyma by fibrous connective tissue. This fibrotic reorganisation of the pancreas leads to progressive exocrine and endocrine pancreatic insufficiency’.1
A recent epidemiological study in France found the annual incidence of chronic pancreatitis was 7.8 per 100,000. Assuming a survival of 15–20 years, the annual prevalence is between 120 to 143 cases per 100,000. Therefore, a GP with a practice of 2,000 patients might expect to have around three cases.2
The most common cause of chronic pancreatitis is alcohol (60-70%), with a logarithmic risk based on exposure.3 However, in approximately 20-30% of cases, the cause is idiopathic and 10% of cases are due other causes. It is especially important to consider other aetiologies in women, as one study found only 28% of cases in women were alcohol-related.4 It is now understood that smoking is an independent and potent risk factor and acts synergistically with alcohol.5 There is also an increasing awareness that obesity and longstanding diabetes predispose to chronic pancreatitis. Other causes to consider include repeated or even a single severe attack of acute pancreatitis and autoimmune pancreatitis (often associated with elevated immunoglobulin G4 serum levels). Numerous genetic mutations and polymorphisms are risk factors and undoubtedly account for many idiopathic cases.6 Cholecystolithiasis alone is not considered a risk factor.
Features
Pain is the hallmark feature of chronic pancreatitis. It is usually dull and epigastric in nature, with radiation to the back. In the early stages pain typically lasts from several hours to several days, but as the disease progresses the attacks become more frequent and pain-free intervals reduce. Not infrequently chronic pancreatitis can be silent, with patients only presenting with the sequelae of exocrine (steatorrhea and weight loss) or endocrine insufficiency (diabetes).
Diagnosis
Symptoms alone are insufficient to diagnose chronic pancreatitis. Instead we rely on structural and functional tests of the pancreas, as well as response to treatment.
The diagnostic difficulty and delay are largely because in the early stage there is often intermittent pain and imaging may be negative. Some bloods tests including low albumin and magnesium can help predict pancreatic insufficiency but are of more use in following patients with established chronic pancreatitis.7 Pancreatic functional tests in the form of faecal elastase-1 (FE-1) are useful especially for diagnosing exocrine insufficiency. A cut off of <200ug/g is commonly used.
The most useful and readily available diagnostic test is a pancreas protocol CT. It will classically show parenchymal atrophy and calcifications, and ductal strictures and dilatations. Abdominal ultrasound should not be used as it lacks sensitivity and ERCP has no diagnostic role due to the risk of complications. Both MRI and endoscopic ultrasound offer higher diagnostic accuracy especially in early disease and should be considered if the CT is non-diagnostic and clinical suspicion is high.8
Management
Chronic pancreatitis requires a long-term management plan. First there needs to be a focus on explaining the diagnosis and building a rapport with the patient. Importantly, avoiding alcohol and tobacco is the key to preventing progression and reducing pain. Next comes the specific management of pain, maldigestion, and local and systemic complications, including diabetes.
Pain
While there are no large randomised controlled trials, the usual approach to pain is to follow the WHO pain ladder incrementally, starting with non-opioids (e.g. paracetamol, ibuprofen), progressing to mild opioids (e.g. codeine, tramadol) and finally, strong opioids (e.g. morphine, oxycodone). Many patients become opioid-dependent and clinicians must watch out for opioid-induced complications such as constipation and narcotic bowel syndrome (a condition where continued or escalating dosages of narcotics actually worsen abdominal pain). In one randomised controlled trial, pregabalin has been found to be modestly effective as an adjuvant therapy for pain.9 In a highly selected group, coeliac plexus block (delivered via endoscopic ultrasound) may be beneficial, although the improvement is often short-lived.
If a patient with established chronic pancreatitis develops new or worsening pain, further cross-sectional imaging is advised, firstly, to exclude complications that mainly comprise pancreatic ductal strictures or stones that can be managed endoscopically with stenting and stone extraction. A smaller proportion may benefit from surgical drainage. Cysts (or pseudocysts) may also arise and cause pain from compressive symptoms and may require drainage. Secondly, the CT can exclude to development of malignancy.
Maldigestion (exocrine insufficiency)
Steatorrhoea (loose, bulky, offensive stools) is synonymous with chronic pancreatitis. It is due to malabsorption (largely of fats) caused by the loss of pancreatic exocrine parenchyma. Treatment is pancreatic enzyme replacement therapy (PERT), the commonest commercially available product being CreonR. Dosage and timing are important, on average 50,000-75,000U are split across each meal with a further 25,000U for snacks. Dosage should be titrated to the relief of symptoms; in some cases, acid suppression with standard PPIs is also beneficial. Once on PERT, patients should be able to tolerate a normal diet, although many patients prefer to eat little and often.
Due to fat soluble vitamin and mineral deficiencies, patients are at higher risk of osteoporosis and therefore should have a bone density assessment every two years.
Diabetes (endocrine insufficiency)
Chronic pancreatitis with the resultant loss of parenchyma and therefore β-cells, often leads to impaired secretion of insulin and the counter-regulatory hormone glucagon. This is called type 3c diabetes and in chronic pancreatitis the lifetime risk is 80%. Patients will often have brittle diabetes and suffer recurrent episodes of hypoglycaemia. Diagnosis is established by measuring HbA1c and fasting glucose which should be checked at least annually. Management incudes addressing malnutrition and PERT and usually starting metformin. Most other oral anti diabetic medications are contraindicated, and many patients progress to requiring insulin.10
Cancer
Chronic pancreatitis is a risk factor for pancreatic cancer, but screening is not effective. The risk of pancreatic cancer in a patient with chronic pancreatitis is estimated to be 1.8% at 10 years and 4% at 20 years.11 In any patient with new symptoms such as increasing pain, weight loss or jaundice, urgent referral for cross sectional imaging should be considered.
In summary chronic pancreatitis is defined by the loss of pancreatic function and usually presents as pain and maldigestion. Treatment is long-term and requires a multidisciplinary team approach to manage; alcohol and tobacco use, pain and its psychological impact, and malnutrition (including advice on how to take PERT) all need to be addressed. Follow up includes surveillance for local (cysts, strictures and malignancy) and systemic complications (diabetes and osteoporosis). As a chronic condition marked with flares of pain, much of this is best accomplished in primary care.
Key points
- On average a GP might expect to care for three patients with chronic pancreatitis a year
- While alcohol is the commonest aetiology, both tobacco and obesity are risk factors and idiopathic and other causes are common
- Diagnosis is often delayed and imaging, even CT, may be normal early in the disease process
- Pain is the predominant symptom and, if new or increasing, warrants cross-sectional imaging to exclude complications (pancreatic ductal obstruction, cysts) or malignancy
- Exocrine insufficiency is best diagnosed by faecal elastase testing and treated with enzyme replacement (PERT)
- Diabetes occurs in up to 80% of cases and requires careful management, especially given coincident malnutrition and the risk of hypoglycaemia
Dr Joe Geraghty is a consultant gastroenterologist at Manchester Royal Infirmary
References
- Hoffmeister A, Mayerle J, Lerch MM. English language version of the S3-consensus guidelines on chronic pancreatitis: Definition, aetiology, diagnostic examinations, medical, endoscopic and surgical management of chronic pancreatitis. Z Gastroenterol. 2015 Dec;53(12):1447-95
- Lévy P, Barthet M, Mollard BR, et al. Estimation of the prevalence and incidence of chronic pancreatitis and its complications. Gastroenterol Clin Biol 2006; 30: 838–844
- Johnson CD, Hosking S. National statistics for diet, alcohol consumption, and chronic pancreatitis in England and Wales,1960-88.Gut,1991,32,1401-1405
- Cote GA, Yadav D, Slivka A, et al Alcohol and smoking as risk factors in an epidemiology study of patients with chronic pancreatitis. Clin Gastroenterol Hepatol 2011;9:266-73.
- Setiawan VW, Pandol SJ, Porcel J, et al . Prospective study of alcohol drinking, smoking, and pancreatitis: the multiethnic cohort. Pancreas 2016;45:819-25.
- Whitcomb DC. Genetic risk factors for pancreatic disorders. Gastroenterology 2013;144:1292-302.
- Lindkvist B, Dominguez-Munoz JE, Luaces-Regueira M, et al. Serum nutritional markers for prediction of pancreatic exocrine insufficiency in chronic pancreatitis. Pancreatology. 2012;12:305-310.
- Lohr JM, Dominguez-Munoz E, Rosendahl J .United European Gastroenterology evidence- based guidelines for the diagnosis and therapy of chronic pancreatitis (HaPanEU). United European Gastroenterology Journal 2017, Vol. 5(2) 153–199
- Olesen S, Bouwense SA, Wilder-Smith OH, et al. Pregabalin reduces pain in patients with chronic pancreatitis in a randomized, controlled trial. Gastroenterology. 2011 Aug;141(2):536-43.
- Rickels MR, Bellin M, Toledo FG, et al. PancreasFest Recommendation Conference Participants. Detection, evaluation and treatment of diabetes mellitus in chronic pancreatitis: recommendations from PancreasFest 2012. Pancreatology 2013;13:336-42.
- Lowenfels AB, Maisonneuve P, Cavallini G, et al. Pancreatitis and the risk of pancreatic cancer. International pancreatitis study group. N Engl J Med. 1993;328(20):1433–7.
Pulse July survey
Take our July 2025 survey to potentially win £1.000 worth of tokens

Visit Pulse Reference for details on 140 symptoms, including easily searchable symptoms and categories, offering you a free platform to check symptoms and receive potential diagnoses during consultations.










