The treatment – neuropathic pain

Neuropathic pain can result from a multiplicity of causes, including peripheral neuropathies, infections and central nervous system disease. Prior to starting therapy for neuropathic pain, the aetiology of the underlying condition needs to be adequately assessed and the pain classified as ‘neuropathic’.
Patients characteristically use certain terms to describe their pain and associated sensory perturbations. There are a number of simple validated and questionnaires that can be used to ascertain these. Our personal preference is for the patient-administered seven items of the DN4, in which a score of greater than 3 suggests a pain of neuropathic origin (see box, right).1 This involves asking the first two questions on symptoms, and scoring each of the seven potential items individually. Although not to be used in isolation, these screening tools are a useful aid to clinical judgment.
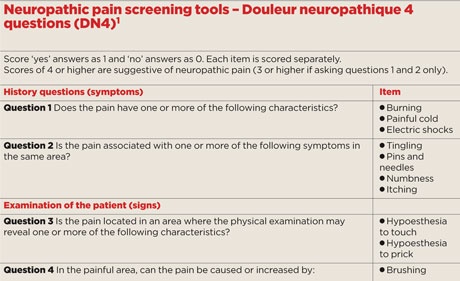
Before starting therapy it is also necessary to document the pain severity so that treatment effect can be monitored at follow-up. Neuropathic pain can occur in three distinct domains even in the same patients, each of which require separate assessment:
• Continuous spontaneous pain – usually best assessed with a simple 0-10 numerical rating scale.
• Paroxysmal pain – best assessed with daily event rate diary.
• Evoked pain.
Neuropathic pain is notoriously challenging to treat, and one of the major research questions is how to stratify patients according to pain mechanisms, and start mechanism-based prescribing for each patient.2 Until this is resolved, there is little option but to continue to approach prescribing on the basis of drugs that have efficacy in clinical trials, adopting a trial-and-error approach until the optimal combination of drugs is found for the individual patient. Regardless of the number of medications previously trialled, it is important to refer patients to a specialist if there is:
• Diagnostic uncertainty.
• Severe pain.
• Pain that significantly limits daily activities.
• The patient’s underlying health condition has deteriorated.3
Pain relief doesn’t appear to follow a normal distribution. It is usually either satisfactory, with a reduction in pain greater than 50% and improvements in associated symptoms such as sleep and fatigue, or it is poor.4 A large number of different therapies have been investigated in randomised controlled trials, but the number of treatments associated with effectiveness is limited.
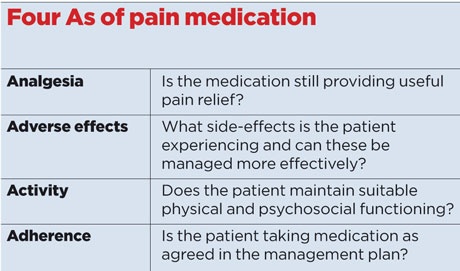
As with all analgesia medication, the principles of managing ongoing therapy include the four As (see box, right). The majority of these pharmacological agents can be used to treat alternative conditions and it is important to stress this to patients prior to initiation. As effective therapies are limited, expectation needs to be appropriately managed.
Standard current treatment
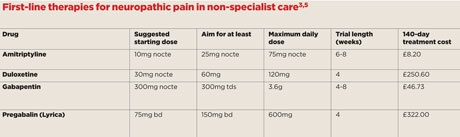
NICE recommends first-line therapies of amitriptyline, duloxetine, gabapentin or pregabalin (Lyrica) for the management of neuropathic pain in non-specialist care, including primary care.3 The key to all of these is gradual titration of the dose to reduce the side-effect profile, and a significant trial length to determine efficacy. If a therapy is not tolerated or not effective after a significant trial, an alternative from the list should be initiated in a similar fashion. All of these analgesia medications are associated with side-effects, so if there are no reported benefits after an adequate trial these should be stopped. When withdrawing or switching treatment, consider tapering the withdrawal with a regimen similar to the titrated trial. Note that guidance issued in March by NHS England now requires prescribers to write the brand name ‘Lyrica’ when prescribing pregabalin for neuropathic pain, and not the generic name.
Antidepressants
Some drugs traditionally used as antidepressants are effective in the treatment of neuropathic pain, independent of any effect on depression.6 The best evidence is for tricyclic antidepressants (TCAs) but there is now promising, although limited, evidence for the role of SSRIs and newer antidepressants such as venlafaxine. Based on the current evidence, treatment should be initiated with amitriptyline and switched to an alternative TCA such as nortriptyline, or an alternative antidepressant such as venlafaxine or duloxetine if some pain relief is achieved but side-effects are troublesome. Effectiveness is usually seen within a few days, but if tolerated a significant trial of four to eight weeks should be started.
TCAs have been shown to be effective in diabetic neuropathy and postherpetic neuralgia and, in small studies, in central pain and atypical facial pain. TCAs and venlafaxine have a similar number needed to treat (NNT) of 3 for moderate pain relief.6 Evidence suggests 60mg and 120mg duloxetine (but not 20mg) are effective for treating pain in diabetic peripheral neuropathy, with a NNT of 6.7 Around 20% of patients discontinue these therapies due to adverse effects, but generally SSRIs are better tolerated than TCAs.6
Gabapentinoids
Evidence supports the use of gabapentin and pregabalin.8 Gabapentin is superior to placebo, providing analgesic benefit in around a third of those with neuropathic pain.9 Adverse effects, including dizziness, somnolence, peripheral oedema and gait disturbance, occur in around two-thirds of patients but are mostly tolerated (only around one in 10 stop due to side-effects). Gradual titration of dose is important to increase tolerance.
Pregabalin at daily doses of 300mg, 450mg and 600mg is effective in postherpetic neuralgia, painful diabetic neuropathy and central neuropathic pain.8 Doses at 150mg daily are generally ineffective. Around 25% of patients stopped treatment due to adverse effects, typically somnolence and dizziness.
Which drug first and in which patient?
There is currently insufficient evidence from direct comparative randomised controlled trials to determine which drug should be used first. Two-drug combinations have been shown to have a superior efficacy, but it isn’t possible to recommend specific combinations.10 Amitriptyline and either gabapentin or pregabalin (Lyrica) may be combined effectively. Combinations of antidepressants are generally not recommended, but if a patient is already taking an SSRI or SNRI for their mood, a trial of a low dose of amitriptyline may be considered (maximum of 25mg).
What’s newly available?
The role of topical therapies is attractive due to their limited side-effect profiles. Localised areas of neuropathic pain may, although evidence is very limited, respond to topical lidocaine 5% plasters (applied for 12 hours within each 24-hour period) or capsaicin cream (0.075% applied four times a day). Examples of localised conditions that may benefit include postherpetic neuralgia or post-surgical neuropathic pain. When prescribing capsaicin cream it is important to emphasise application precautions and warn patients that two to six weeks of regular application may be required before maximum analgesic benefit is achieved.11
Many patients stop capsaicin treatment due to localised burning discomfort. As data are insufficient it is difficult to draw conclusions regarding the efficacy of low-concentration capsaicin cream or lidocaine 5% plasters.11, 12
What is out of fashion and why?
There are significant risks associated with long-term opioid use (including tramadol) and therefore these risks should be discussed at initiation, and such therapy only initiated in collaboration with pain medicine specialists.13, 14 Evidence of the effectiveness of opioids in the long-term management of chronic pain is limited but there is increasing evidence regarding their adverse effects and associated morbidity and mortality – up to 80% of patients taking opioids will experience adverse effects.13, 15
Many patients discontinue long-term opioid therapy due to adverse effects or insufficient pain relief. Long-term opioids, including tramadol, should only be prescribed in exceptional circumstances and in accordance with the British Pain Society and RCGP’s Opioids for persistent pain: good practice guide.13
For neuropathic pain, excluding trigeminal neuralgia, there is insuf?cient evidence for antipsychotics, valproic acid or sodium valproate, phenytoin, oxcarbazepine, lacosamide, lamotrigine, clonazepam and vitamin D.16-24
Special cases
Trigeminal neuralgia is managed differently from other types of neuropathic pain. Expert advice should be sought early as management of this condition is highly specialised and there may be a role for intervention and surgery.
Carbamazepine is probably an effective therapy and first-line treatment. Start at 100mg twice daily, aiming to titrate to at least 200mg four times a day (maximum of 1.6g daily).25
Alternative treatment options include oxcarbazepine, baclofen and lamotrigine. There is currently no good long-term (longer than four weeks) evidence for the effectiveness of carbamazepine in the management of chronic neuropathic pain. It is probably effective in some people, but on current data caution should be maintained.
Key points
• NICE recommended first-line treatments for non-specialist use include amitriptyline, duloxetine, gabapentin and pregabalin (Lyrica). Titration of dose and an effective length of trial are important.
• Long-term opioids (including tramadol) are not known to be effective in managing neuropathic pain and are associated with significant adverse effects.
• The role of physical interventions, including neuromodul-ation, in neuropathic pain is not supported by robust evidence.
Professor Andrew Rice is an honorary consultant in pain medicine at Chelsea and Westminster Hospital NHS Foundation Trust, and professor of pain research at Imperial College London.
Dr Ruth Cowen is an advanced pain trainee at Chelsea and Westminster Hospital NHS Foundation Trust.
References
1 Bouhassira D, Attal N, Alchaar H et al. Comparison of pain syndromes associated with nervous or somatic lesions and development of a new neuropathic pain diagnostic questionnaire (DN4). Pain 2005;114:29-36
2 Baron R, Forster M, Binder A. Subgrouping of patients with neuropathic pain according to pain-related sensory abnormalities: a first step to a stratified treatment approach. Lancet Neurol 2012;11:999-1005
3 NICE clinical guideline 173. Neuropathic pain – pharmacological management (The pharmacological management of neuropathic pain in adults in non-specialist setting).November 2013
4 Moore A, Derry S, Eccleston C. Expect analgesic failure; pursue analgesic success that works for each patient. BMJ 2013;2690:7-9
5 Map of medicine Neuropathic pain.
6 Saarto T, Wiffen PJ. Antidepressants for neuropathic pain (Review). Cochrane Database Syst Rev 2007 Oct 17;(4)
7 Lunn MP, Hughes RA, Wiffen PJ. Duloxetine for treating painful neuropathy or chronic pain (Review). Cochrane Database Syst Rev 2014 Jan 3;1
8 Wiffen PJ, Derry S, Moore RA et al. Antiepileptic drugs for neuropathic pain and fibromyalgia – an overview of Cochrane reviews (Review). Cochrane Database Syst Rev 2013 Nov 11;11
9 Moore RA, Wiffen PJ, Derry S, et al. Gabapentin for chronic neuropathic pain and fibromyalgia in adults (Review). Cochrane Database Syst Rev 2014 Apr 27;4
10 Chaparro LE, Wiffen PJ, Moore RA et al. Combination pharmacotherapy for the treatment of neuropathic pain in adults (Review). Cochrane Database Syst Rev 2012 Jul 11;7
11 Derry S, Moore RA. Topical capsaicin (low concentration) for chronic neuropathic pain in adults (Review). Cochrane Database Syst Rev 2012 Sep 12;9. 22
12 Khaliq W, Alam S, Puri N. Topical lidocaine for the treatment of postherpetic neuralgia (Review). Cochrane Database Syst Rev 2007 Apr 18;(2).23
13 The British Pain Society. The British Pain Society’s Opioids for persistent pain: Good practice; 2013
14 McNicol ED, Midbari A, Eisenberg E. Opioids for neuropathic pain (Review). Cochrane Database Syst Rev 2013 Aug 29;8
15 Noble M, Treadwell JR, Tregear SJ et al. Long-term opioid management for chronic noncancer pain (Review). Cochrane Database Syst Rev 2010 Jan 20;(1)
16 Seidel S, Aigner M, Ossege M et al. Antipsychotics for acute and chronic pain in adults (Review). Cochrane Database Syst Rev. 2013 Aug 29;8
17 Gill D, Derry S, Wiffen PJ et al. Valproic acid and sodium valproate for neuropathic pain and fibromyalgia in adults (Review). Cochrane Database Syst Rev 2011 Oct 5;(10)
18 Birse F, Derry S, Moore, RA. Phenytoin for neuropathic pain and fibromyalgia in adults (Review). Cochrane Database Syst Rev 2012 May 16;5
19 Zhou M, Chen N, He L et al. Oxcarbazepine for neuropathic pain (Review). Cochrane Database Syst Rev 2013 Mar 28;3
20 Derry S, Gill D, Phillips T et al. Milnacipran for neuropathic pain and fibromyalgia in adults (Review). Cochrane Database Syst Rev 2012 Mar 14;3
21 Hearn L, Derry S, Moore RA. Lacosamide for neuropathic pain and fibromyalgia in adults (Review). Cochrane Database Syst Rev 2012 Feb 15;2
22 Wiffen PJ, Derry S, Ra M. Lamotrigine for chronic neuropathic pain and fibromyalgia in adults (Review). Cochrane Database Syst Rev 2013 Dec 3;12
23 Corrigan R, Derry S, Wiffen PJ et al. Clonazepam for neuropathic pain and fibromyalgia in adults (Review). Cochrane Database Syst Rev 2012 May 16;5
24 Straube S, Derry S, Moore RA et al. Vitamin D for the treatment of chronic painful conditions in adults (Review). Cochrane Database Syst Rev 2010 Jan 20;(1)
25 Wiffen PJ, Derry S, Moore RA et al. Carbamazepine for chronic neuropathic pain and fibromyalgia in adults (Review). Cochrane Database Syst Rev 2014 Apr 10;4
Visit Pulse Reference for details on 140 symptoms, including easily searchable symptoms and categories, offering you a free platform to check symptoms and receive potential diagnoses during consultations.













