Does a rash while on antibiotics mean an allergy to that drug?
What is the myth and where did it come from?
Allergy is one of the most confusing and poorly understood topics in medicine. Even immunologists don’t fully understand it. Anyone wanting to pick me up on that outlandish statement is welcome to write an email of complaint, as long as they also explain why the advice on peanut exposure to pregnant mothers and babies has changed so many times over the years.
The complexities and misunderstandings relating to allergy offer the perfect environment for the perpetuation of myths. It is far easier to resolve a myth if the subject matter is simple.
A basic aspect of allergy that is often not fully understood is that there are two types – IgE and non-IgE. If someone grabs you and says, ‘Come quickly – someone is having an allergic reaction!’ you’ll probably find it is caused by an IgE-type allergy. If they are having a stomach cramp because of bread they ate an hour earlier (a non-IgE reaction), you’ll be unimpressed at being dragged away from what you were doing. Anaphylaxis is an extreme IgE type reaction, but many people with food allergies have non-IgE reactions, mainly gastrointestinal symptoms.
Poor understanding of this distinction allows misleading statements such as: ‘Fatal food anaphylaxis for a food-allergic person is rarer than accidental death in the general population’.1 Note, this does not distinguish between IgE and non-IgE allergy.
A key reason so many myths arise around allergy is that we are all terrified of the potential consequences. Clinicians have a healthy respect for drug allergy, partly through anxiety about anaphylaxis and partly because we don’t want to be the direct cause of harm to our patients. There are so many myths about antibiotic allergy that it can be hard to know what the truth is. Most of us, if there is any uncertainty, will choose what seems like the safest option. For example, clinicians sometimes avoid prescribing penicillin where there is a family history of penicillin allergy.2 But inheritance is not an established factor in this case. Penicillin allergy does not truly ‘run in families’ but is so common a diagnosis that more than one family member might have been given it. Therefore it is usually safe to prescribe despite such a family history.
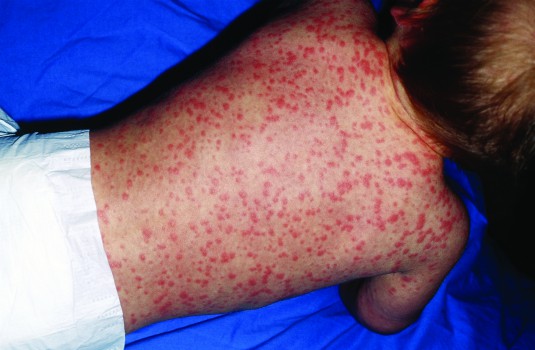
For a long time there has been a huge problem with overdiagnosis of antibiotic allergy in children. If a child gets an urticarial rash while taking penicillin, this is often presumed to be the result of an allergy. This is a myth many allergy specialists would like to bust and it is built upon other myths. One of these is that if the reaction was simple urticaria, a subsequent reaction could be anaphylaxis. Again, this is not backed up by sound evidence.
What is the reality?
It is well known that only about 5% of people who believe themselves to be allergic to penicillin truly are.3 Often a patient mistakenly thinks they are allergic when in fact they have suffered a side-effect of the antibiotic. However, in many cases, patients have been given the antibiotic allergy label by a healthcare professional. In children, the most common reason for this incorrect label is that rashes and wheezes are extremely common in the context of a paediatric viral illness. When a child develops a rash while taking antibiotics, the chances are that the rash is due to the illness, not the drugs. This is true for urticaria just as it is for a standard macular-papular non-specific rash.
Of course there is the possibility the child does have a true antibiotic allergy so it is assumed the safe thing to do is to advise avoidance. This is especially so given the fears that future use will lead to much worse reactions, although the evidence suggests this is unlikely to be true.3 Studies that have challenged patients with a label of penicillin allergy, have often done so using a standard oral dose of the antibiotic in question. In my favourite study,3 this brought no reaction in 95% of subjects – but more importantly, those who did react experienced symptoms no worse than those that originally resulted in their being labelled allergic.
Indeed, I am repeatedly told by allergy specialists that there is no evidence to suggest repeated exposure to an allergen increases the severity of an allergy once it is established. What can worsen the reaction is increasing the dose or altering the route (for instance, intravenous instead of oral). If the dose and route are unchanged, the evidence is that the reaction will be the same.
There is even an additional myth that there is a 10% chance of a cross-reaction with cephalosporins if a child has a diagnosis of penicillin allergy. In fact, that figure is an overestimate and only applies to earlier-generation cephalosporins.4
How does this change my practice?
There are several ways that dispelling these myths could lead to changes in practice. The first is that we could all stop the presumptive approach to diagnosing an antibiotic allergy in a child. If a child given antibiotics for a respiratory tract infection develops a non-specific or urticarial rash with no other signs of an allergic reaction, it is reasonable to attribute the rash to a viral cause. The published evidence tells us we will be right in the vast majority of cases. More importantly, if the child has had a true antibiotic allergy, if they are given the same antibiotic again in a normal dose by the oral route, the next reaction should be a similar one to the event they have just had.
The really big opportunity here is the ability to correct a child’s diagnosis of antibiotic allergy. GPs are increasingly doing this on the basis of evidence-based advice from allergy specialists. Removal of the label then allows the child to be prescribed the most appropriate antibiotic – effectively challenging the initial allergy diagnosis.
How this challenge is carried out varies from clinician to clinician. Some take the view that if there is no real risk, it is best to give a full explanation to the parents and advise them to give the child the antibiotic at home and seek reassessment if the rash recurs. Parents who are anxious about administering the first dose can be invited to do this in the surgery and then stay in the waiting room for an appropriate amount of time.
These undiagnosing strategies are only suggested for children where the history is of rash without oral swelling or wheeze. It is also only recommended for low-risk patients. Experts have suggested that challenging outside of secondary care should not be done for children with asthma, food allergies or a history of anaphylaxis to another allergen.
Finally, this is yet another reason not to prescribe antibiotics too easily. When they are simply an option, rather than being truly needed, they are usually best avoided. All this diagnosing and undiagnosing of antibiotic allergy is a hassle that we could do without and we will face it a lot less often if we have an appropriate threshold for prescribing antibiotics in the first place.
Edward Snelson is a consultant paediatrician specialising in paediatric emergency medicine at the Sheffield Children’s Hospital. He is the author of The Essential Clinical Handbook of Common Paediatric Cases and of gppaedstips.blogspot.co.uk – a website for GPs who want to keep up to date with paediatrics in primary care. Before becoming a paediatrician, he was a GP for more than five years, and now uses that experience to fuel his educational work in primary care.
Twitter: @sailordoctor
References
- Umasunthar T et al. Incidence of fatal food anaphylaxis in people with food allergy: a systematic review and meta-analysis. Clinical and Experimental Allergy, 2013;43:1333-41 dx.doi.org/10.1111/cea.12211
- American Academy of Allergy, Asthma and Immunology. Penicillin Allergy FAQ. tinyurl.com/AAAAI-penicillin
- Caubet J et al. The role of penicillin in benign skin rashes in childhood: a prospective study based on drug rechallenge. J Allergy Clin Immunol 2011;127:218-22
- Bhattacharya S. The Facts About Penicillin Allergy: A Review. J Adv Pharm Technol Res 2010; 1:11–17
Visit Pulse Reference for details on 140 symptoms, including easily searchable symptoms and categories, offering you a free platform to check symptoms and receive potential diagnoses during consultations.













