Everything GPs need to know about identifying and managing Strep A
Dr Toni Hazell provides a comprehensive guide for GPs managing Strep A in primary care
It has been a tough few years in primary care – as we reach the end of 2022, COVID-19 is still around and we are all struggling with unprecedented demand in primary care. So it was just what we needed to hear about another infectious agent causing a spike of demand for healthcare, panic among the general public and shortages of key medications. Merry Christmas.
What is iGAS and how many cases are there?
So, what is this acronym iGAS? It stands for invasive Group A Streptococcus. Group A Streptococcus (GAS) is carried as a commensal in the nasopharynx of 5 – 15% of the population1 and can cause pharyngitis and scarlet fever. iGAS is a rare but serious complication of infection with GAS and occurs when the bacteria infects a site that is usually sterile such as the blood, joints or lungs2. At the time of writing, 15 deaths of UK children have been linked to iGAS3. The greatest increase is in those aged 1 – 9, with much smaller increases in those aged under 1 or 10 and over, as shown in the table below – these numbers might also be useful to help to put the absolute risk in perspective for parents i.e. only an extra two cases or less per 100,000 children4. This year there have been more cases of GAS in the lower respiratory tract, including cases of empyema, and more cases of scarlet fever, which is a notifiable disease2.
Age iGAS average case numbers per 100,000 children Pre-pandemic (2017 – 2019) iGAS average case numbers per 100,000 children December 2022 Under 1 1.1 1.3 1 – 4 0.5 2.3 5 – 9 0.3 1.1 10 – 14 0.1 0.2
Why are we seeing this increase now?
GAS is spread in the same way as other respiratory viruses, by close contact and airborne droplet spread. If it infects the skin (it can cause cellulitis and impetigo) then spread can also be from a wound. There doesn’t appear to be a new circulating strain of GAS5 and it is likely that there are a number of factors contributing to high amounts of circulating bacteria. This could include increased social mixing, after the years of the pandemic where this didn’t happen, as well as higher levels of other respiratory tract infections. A breach in the epithelium of the respiratory tract following an infection such as flu can enable GAS to enter the lungs, causing iGAS5. iGAS can only really be confirmed following secondary care investigations, such as blood cultures; our role in primary care isn’t to diagnose iGAS specifically but to assess for the risk of GAS and treat appropriately, as well as to spot the occasional child who is unwell enough that it is a serious consideration, and refer them to hospital.
How can we triage effectively and efficiently?
So, the million dollar question is – how can we in primary care safely triage the vast numbers who are worried about Strep and want to be seen and see them all in an efficient manner, while not missing the needle in a haystack case of iGAS? I don’t think I have an answer to the first part of that as systems will vary locally. GPs who usually do phone triage seem to have generally decided to lower their threshold for seeing children with symptoms of respiratory tract infection face to face at the moment and that seems like a sensible move. The development of iGAS from early respiratory symptoms is very difficult to predict but seeing face to face must surely increase that chance, as well as providing medicolegal protection if things go wrong.
Seeing everyone face to face saves time on triage, but takes more time on face-to-face appointments and it will be up to individual practices how they manage it. Do you get a nurse or HCA to see them first and do observations? Do you give appointments or have a sit-and-wait system? What resources do you have locally if demand outstrips what you can reasonably cope with, and is your ICB sympathetic to the current pressures? In some areas overflow capacity has been established to support GPs, whereas in others the response has been more in the form of vaguely threatening emails sent to practices who have a higher proportion of children attending A&E.
What tools do we have at our disposal in face-to-face consultations?
Moving on from the population wanting to be seen, what should we be looking for when we have a patient and their concerned parent in front of us? The first look at a child is always important. I always prefer not to use the tannoy, but instead to go out into the waiting room and call the patient. If a child has to be caught, because they are running around pretending to be an aeroplane, or they have to be peeled away from a massive packet of crisps that they are halfway through, that’s reassuring. Conversely, I have occasionally spotted a clearly unwell child lying in their parent’s arms and moved them up the appointment list.
Never ignore your ‘spidey sense’ or gut feeling that a child is ill – one study has shown that this intuition is associated with an increased likelihood of severe illness and that acting on it has the potential to prevent unwell children being missed6. If we want to define that in a more systematic way then we could use the NICE guidance on fever in under 5s7 which reminds us of general features to look out for, including a child who is pale or mottled, doesn’t respond to social cues, needs regular rousing, has reduced skin turgor or skin indrawing or is tachypnoeic.
Green – low risk Amber – intermediate risk Red – high risk Colour (of skin, lips or tongue) Normal colour Pallor reported by parent/carer Pale, mottled, ashen or blue Activity • Responds normally to social cues
• Content or smiles
• Stays awake or awakens quickly
• Strong normal cry or not crying • Not responding normally to social cues
• No smile
• Wakes only with prolonged stimulation
• Decreased activity • No response to social cues
• Appears ill to a healthcare professional
• Does not wake or if roused does not stay awake
• Weak, high-pitched or continuous cry Respiratory • Nasal flaring
• Tachypnoea: respiratory rate
• >50 breaths per minute, age 6 to 12 months;
• >40 breaths per minute, age more than 12 months
• Oxygen saturation less than or equal to 95% in air
• Crackles in the chest • Grunting
• Tachypnoea: respiratory rate more than 60 breaths per minute
• Moderate or severe chest indrawing Circulation and hydration • Normal skin and eyes • Moist mucous membranes • Tachycardia:
• More than 160 beats per minute, age less than 12 months
• More than 150 beats per minute, age 12 to 24 months
• More than 140 beats per minute, age 2 to 5 years
• Capillary refill time more than or equal to 3 seconds
• Dry mucous membranes
• Poor feeding in infants
• Reduced urine output • Reduced skin turgor Other • None of the amber or red symptoms or signs • Age 3 to 6 months, temperature more than or equal to 39°C
• Fever for more than or equal to 5 days
• Rigors
• Swelling of a limb or joint
• Non-weight bearing limb or not using an extremity • Age less than 3 months, temperature more than or equal to 38°C
• Non-blanching rash
• Bulging fontanelle
• Neck stiffness
• Status epilepticus
• Focal neurological signs
• Focal seizures
The NICE guidance on sepsis also covers older children and adults8 and reminds us that symptoms may be non-specific, that not everyone with sepsis has a high temperature and that we should be particularly careful when assessing those who have communication difficulties, due to language or other reasons. The NEWS2 score9 is recommended by NHSE for use across ambulance services and secondary care, but it is not validated in primary care and the RCGP has raised concerns about it being requested from primary care, suggesting that we should instead be able to provide physiological measurements (pulse, temperature etc) but that NEWS2 should not be mandated in primary care unless it is validated in this setting by further research10.
What are the clinical presentations we might encounter?
Clinical presentations of GAS may include a simple sore throat, tonsillitis or scarlet fever. Scarlet fever, which is a notifiable disease, presents in a typical way. The initial symptoms are non-specific (sore throat, headache, fever) and a rash will develop within two days. The rash is rough, described as like sandpaper, and it starts on the torso and spreads from there, but usually spares the palms and the soles. Pastia’s lines may be seen – these are deep red lines where the rash is accentuated in the flexures11.
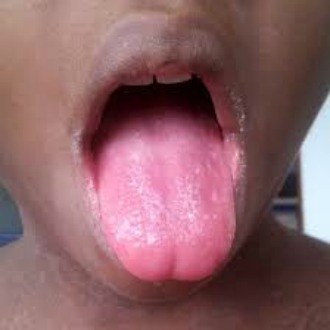
Other features include a strawberry tongue – the tongue has a white coating which peels off after a few days, leaving the tongue looking red and swollen – enlarged cervical lymph nodes, general malaise, peeling skin on the fingertips and toes and a flushed face with pallor around the mouth2. Patients with scarlet fever who aren’t ill enough to need admission should be treated with 10 days of oral penicillin V11, but if that isn’t available then other options include amoxicillin, macrolides and cefalexin, in decreasing order of preference2. Macrolides are the first choice for those with penicillin allergy2. Five days of antibiotics will be appropriate for many children, but this is at the discretion of the treating clinician and we should be aware that 10 days increases the chance of microbiological cure1.
For those who have a sore throat or tonsillitis, but no signs of scarlet fever, and who are not so ill that you feel they need hospital review (using the tools already mentioned if needed to aid that judgment), the decision has to be made as to whether they need antibiotics. The NICE guidance on sore throat recommends two decision making tools – Centor and FeverPAIN12 but the interim clinical guidance summary produced by NHSE, the UKHSA, the RCGP, the RCPCH, the Royal Pharmaceutical Society and NICE seems to focus on FeverPAIN because it is validated in primary care and suggests that we adjust our threshold for antibiotic prescribing to prescribe for those with a FeverPAIN score of 3 or more2.
FeverPAIN/Centor criteria
FeverPAIN criteria
• Fever (during previous 24 hours)
• Purulence (pus on tonsils)
• Attend rapidly (within 3 days after onset of symptoms)
• Severely Inflamed tonsils
• No cough or coryza (inflammation of mucus membranes in the nose)
Each of the FeverPAIN criteria score 1 point (maximum score of 5). Higher scores suggest more severe symptoms and likely bacterial (streptococcal) cause. A score of 0 or 1 is thought to be associated with a 13 to 18% likelihood of isolating streptococcus. A score of 2 or 3 is thought to be associated with a 34 to 40% likelihood of isolating streptococcus. A score of 4 or 5 is thought to be associated with a 62 to 65% likelihood of isolating streptococcus.
Centor criteria
• Tonsillar exudate
• Tender anterior cervical lymphadenopathy or lymphadenitis
• History of fever (over 38 degrees Celsius)
• Absence of cough
Each of the Centor criteria score 1 point (maximum score of 4). A score of 0, 1 or 2 is thought to be associated with a 3 to 17% likelihood of isolating streptococcus. A score of 3 or 4 is thought to be associated with a 32 to 56% likelihood of isolating streptococcus.
This is a significant lowering of the threshold as the NICE guidance on sore throats usually advises only that we consider an immediate or back-up antibiotics with a FeverPAIN score of 4 or 5 and that we consider a back-up antibiotic or no antibiotic with a score of 2 or 3. In normal times, a FeverPAIN score of 2-3 is associated with a 34-40% likelihood of isolating streptococcus and a score of 4-5 is associated with a 62 – 65% chance, however increased incidence may mean that these predictive values are increased at the current time. The FeverPAIN score has been criticised for giving one point for those who attend within three days of the onset of symptoms. This is presumably meant to indicate severity of symptoms, however in an environment of public panic and the trend towards wanting immediate access to a doctor for all respiratory symptoms, it is arguable whether attending quickly is actually a marker of severity.
Should I take a throat swab?
Throat swabs do not usually play a significant role in the management of sore throat, or the investigation of scarlet fever11 in the community – Strep A is a commensal, so its presence doesn’t necessarily mean that it is the cause of a sore throat, and the results aren’t back until some days after the decision on whether to treat has to be made. The current guidance says that we should consider taking a throat swab where there is ‘diagnostic uncertainty’ or concerns about antibiotic resistance – presumably so that if first-line antibiotics don’t help, we have sensitivities to guide the second choice. However, in some areas swabbing may be arranged as part of public health investigations – this is in line with NICE CKS advice on scarlet fever which says that we should consider swabbing if ‘a case is suspected to be part of an outbreak — the local health protection team should advise primary care if a local outbreak is suspected and when testing is appropriate’.
The same guidance also suggests swabbing if there is a true allergy to penicillin and if a case is in regular contact with vulnerable people, for example healthcare workers. However neither of these last two suggestions have been specifically made in relation to the current outbreak and so swabbing should probably be reserved for when there is diagnostic uncertainty, or if asked to do so by public health.
What can I do when liquid antibiotics are not available?
With an increase in the number of children being seen for respiratory symptoms, and a lowered threshold for antibiotic prescribing, it comes as no huge surprise that the price of antibiotics has increased13 and availability has become an issue14, particularly of the liquid preparations. A useful article from the Specialist Pharmacy Service15 gives some practical tips that can be used when liquid antibiotics are not available. These include online resources to show older children how to swallow tablets (a useful life skill which not all adults have yet acquired), and advice on how to crush tablets or open capsules. Use of medicines in this way is off-licence, but may be necessary if liquid forms cannot be obtained.
It would be sensible not to send the script to a nominated pharmacy but instead give a paper script or upload to the spine for any pharmacy to download, to avoid the prescription being held in a pharmacy that doesn’t have stock. For the same reason give the antibiotic script on a separate piece of paper to any other scripts.
What are the rules about isolation and prescribing for contacts?
Parents may ask us whether their children with scarlet fever or a sore throat should isolate or stay off school – there have been no changes in the law or new rules issued, as happened during the pandemic, but we do have a pre-existing document16 which outlines how long children should stay off school/nursery for various common infections, which was last updated on 7 December. It advises that a person is infectious for 2-3 weeks if antibiotics are not used, but that exclusion from school or nursery need only be for 24 hours after starting antibiotics. In a similar vein, you may be asked to prescribe antibiotics for family members, or other contacts of a child with a Strep related infection. This should really be the remit of public health both to make the decisions and to provide the prescriptions, as it is not contractually the role of general practice to provide antibiotic prophylaxis for those who are not ill – this has been shown by the development of enhanced services to do this when there are flu outbreaks in care homes17.
It has been suggested2 that antibiotic prophylaxis may be needed for close contacts of a case who are pregnant or post-natal (≥ 37 weeks or within one month of delivery), aged < 28 days or ≥ 75 years, or who develop chickenpox within a week before or two days after exposure to iGAS. A close contact is defined as prolonged contact in a household-type setting during the seven days before the onset of symptoms and up to 24 hours after initiation of appropriate antimicrobial therapy.
We don’t know how long this outbreak will continue and whether advice will change rapidly, or stay the same for weeks. As with any medical issue, the key principles should remain the same. Take a good history including eliciting the concerns of the patient/parent, carry out a thorough examination, document your findings carefully and safety-net for any future changes. A child who is completely well when seen by their GP might later develop iGAS and we can’t prevent that, but we can do our best to practise safely so that we have a chance of seeing that needle glinting away in the middle of the haystack.
Dr Toni Hazell is a GP in North London
References (all viewed 11.12.22)
Visit Pulse Reference for details on 140 symptoms, including easily searchable symptoms and categories, offering you a free platform to check symptoms and receive potential diagnoses during consultations.
Related Articles
READERS' COMMENTS [6]
Please note, only GPs are permitted to add comments to articles















Solve the antibiotic shortage and dump the guesswork by using £3 rapid result Strep A throat swabs.
Any chance of the NHS joining the 21st century?
Not sure how useful that is though – Strep A is a commensal so you presumably wouldn’t want to treat everyone with a positive swab. What’s the specificity and sensitivity of the rapid swabs? Risk is that we just look at the swab and stop looking at the patient.
Strep A is laughing at human hysteria.
Rapid testing for Strep is only useful in a private Healthcare setting where profit is the motive. This is because its main purpose is to drive up demand and hence turnover. In the NHS it would not change the treatment since the treatment is based on the clinical assessment. You will simply drive up demand for people needing to be “checked out”. In the US it’s main purpose is to increase the throughput of worried well which have a high profit margin.
A negative rapid Strep A test is a considerable relief for anxious parents, whether that anxiety is entirely rational or not.
A negative rapid Strep A test means you can convincingly justify not prescribing yet another course of unnecessary amoxil.
“Rapid testing for Strep is only useful in a private Healthcare setting where profit is the motive . .” They used to say that about Laparoscopy; how did that work out?
“What’s the specificity and sensitivity of the rapid swabs?”. Would have to be pretty useless to be worse than CENTOR scoring, which is guesswork in a white coat.
FeverPain is even worse
My kids got a temp, and rapid access to GP due to the mass hysteria – already scores 2 on fever pain
So medium risk, requiring antibiotics??