GPs asked to administer Covid boosters in care homes by 1 November, ideally with flu
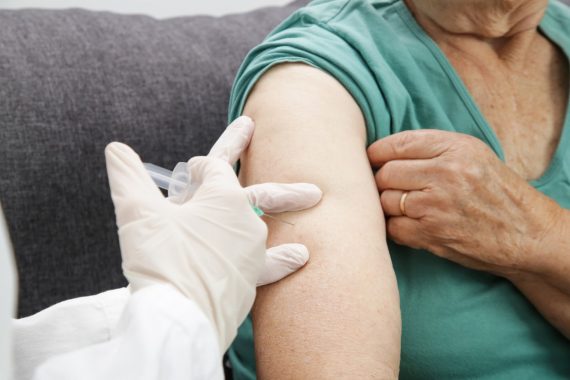
GPs have been told to prioritise offering Covid boosters to care home patients and staff by 1 November and are encouraged to co-administer the vaccine with the flu jab.
NHS England said a ‘short delay’ in delivering the flu jab in order to give it alongside the Covid vaccine could help ensure more people receive both vaccines in a care home setting.
New guidance on the booster programme published today said that GP-led vaccination sites should ‘prioritise care homes for older adult care home residents and care home staff’ – the first priority cohort recommended for boosters.
All eligible patients in the cohort should be offered a vaccination by 1 November and ‘delivery plans should be designed to meet this target’, it added.
The guidance said that GPs should ‘make preparations to start vaccinating as soon as possible’ and that NHS England expects to open the national booking system and issue first national invitations ‘based on available capacity’ on Monday.
NHS England reiterated JCVI guidance that Covid and flu vaccines can be co-administered ‘where operationally expedient’ but that the booster programme ‘should [not] disrupt or delay deployment’ of the flu programme.
However, it said that GPs should ‘use their discretion’ to delay flu jabs for co-administration if this would boost uptake.
The letter said: ‘It is important individuals are offered their Covid-19 and influenza vaccine as soon as they are eligible, rather than delaying for the purpose of co-administration.
‘[But] we recognise there will be some instances where a short delay will ensure that more individuals receive both vaccines, for example in care homes, and sites should use their discretion to maximise these opportunities.’
It added that GPs should ‘consider co-administration wherever eligibility for both programmes, supply and regulation allow’ and ‘seek to co-administer in any instances where it improves patient experience and uptake of both vaccines, reduces administrative burdens on services or to reduce health inequalities (eg in HHs, residential care homes and roving models).’
And NHS England said that GPs should ‘take every opportunity to promote the uptake of both vaccination programmes, including booking individuals in for their influenza vaccine in instances where co-administration cannot realistically be done [and] taking into account patient choice where eligible patients wish to receive their vaccinations at separate appointments’.
They should also use appointments for ‘other health promotion activity’ where ‘practical and appropriate’, it added.
Practices’ collaboration agreement will be updated to support ‘practices working as part of a PCN grouping in using their supply of seasonal influenza vaccine at Covid-19 vaccine designated sites’, for example at ‘joint clinics’, NHS England said.
NHS England will also update the phase three enhanced service specification and standard operating procedures, it added.
Practices will be eligible for an additional payment of £10 per dose on top of the item of service (IOS) fee for all vaccines delivered in a care home setting, in line with previous phases of the vaccination programme, NHS England said.
And financial support for PCN clinical directors due to expire this month will be extended until March 2022, although it will be reduced ‘to ensure it is affordable within the overall funding envelope for the vaccination programme’, it added.
NHS England said: ‘In recognition of the valuable work carried out by PCNs, from October 2021, this funding will be equivalent to an increase in clinical director time per PCN from 0.25 WTE to 0.75 WTE for this period. The payment is to support the leadership and management of the Covid-19 support.’
The guidance also said:
- NHS England will issue further advice on delivering half-doses of the Moderna vaccine but does not expect to go live with this immediately
- There are no supply constraints and PCN-led sites will be able to order vaccine supply as needed through the Foundry ordering platform
- Point of care systems have been updated so that booster vaccines can be recorded
- Until the patient group direction (PGD) and national protocol have been updated – expected by 21 September and ‘shortly after’ respectively – only sites with a prescriber on-site can deliver booster jabs
- Hospital-led sites will be the ‘default provider’ of booster jabs for eligible health and social care staff although local systems ‘may have developed alternative local arrangements’ with GP-led sites
- Hospital hubs will also deliver opportunistic booster vaccines to inpatients and outpatients
The Government yesterday accepted final JCVI advice that all patients in groups 1-9 of the first phase of the Covid vaccination campaign should have a booster jab six months after their second dose, prioritised in the same order as in phase one.
The JCVI recommended administering a full dose of the Pfizer vaccine, in the first instance, or half dose of the Moderna vaccine as an alternative – with the Astra Zeneca vaccine considered in cases where a patient has an allergy to Pfizer and Moderna.
At the same time, medicines regulator the MHRA determined that co-administration of flu and Covid jabs is safe and can go ahead.
Health secretary Sajid Javid told MPs yesterday afternoon that the booster jab rollout will begin ‘next week’ and deputy chief medical officer (CMO) Professor Jonathan Van Tam said he expects the NHS to be able to begin within ‘days’.
Pulse July survey
Take our July 2025 survey to potentially win £1.000 worth of tokens

Visit Pulse Reference for details on 140 symptoms, including easily searchable symptoms and categories, offering you a free platform to check symptoms and receive potential diagnoses during consultations.
Related Articles
READERS' COMMENTS [6]
Please note, only GPs are permitted to add comments to articles











Sorry no can do, too busy seeing patients f2f as per the health secretary yesterday….
Absolutely Michael, always love to hear such helpful advice from the experts, especially experts who are not currently at the coal face and leaders who have never been there??
‘GPs have been told to prioritise offering Covid boosters to care home patients and staff by 1 November and are encouraged to co-administer the vaccine with the flu jab.’
It’s hilarious being told to ‘PRIORITISE’ different things on consecutive days by philosiphising ‘humanities trained folk’ (tfic)
– ignorant ill-informed arseholes – Oops, didn’t mean to say that…
just say no–let hospitals cover
Tbh a nurse or cleaner could do it. Give them the due accolades.
Decorum Est——–careful—-the censors are very sensitive these days