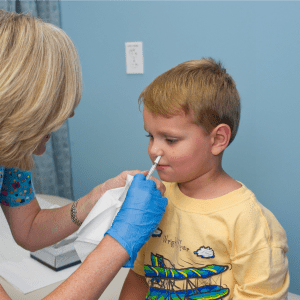
GP practices can go ahead and order child flu vaccine as normal despite an unexpected shortage, Public Health England has said.
Some of the pilots for administering the nasal vaccine to school children will be halted while a supply issue is resolved.
PHE said the situation had arisen because some of the vaccine that had been intended for use had not met ‘strict licensing specifications’.
However, a spokesperson told Pulse that GP practices could go ahead and order vaccine stocks as planned, saying there should be ‘no effect on GP ordering whatsoever’.
The spokesperson added that while vaccination pilots for school children aged 7-10 will be put on hold until new stock arrives from the US, the school vaccinations for children aged 5-6 will go ahead as planned.
GPs are responsible for vaccinating children aged 2-4.
PHE medical director Professor Paul Cosford said: ’In recent days, AstraZeneca, the vaccine’s manufacturer, has informed us of a supply issue. Because some of the vaccine did not meet the strict licensing specifications, it will not be used.
‘As with all of our vaccination programmes, we are using an established contingency plan and AstraZeneca are currently putting an alternative supply of the same vaccine from the US through European testing. Providing they meet the standards, these batches will be made available in the coming weeks.’
Pulse October survey
Take our July 2025 survey to potentially win £1.000 worth of tokens