GPs face immediate cap on child flu vaccine orders
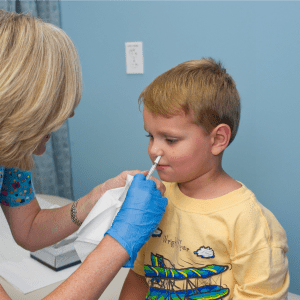
GP practices will only be able to order a maximum of 50 doses of nasal flu vaccine next week to start routine immunisations in children, public health officials have warned.
Public Health England (PHE) said it will be temporarily capping orders of the new Fluenz Tetra vaccine at 50 doses – or five packs – per practice per week, when central ordering of the vaccine opens next Wednesday, 24 September.
PHE said in its latest vaccine update the cap was needed to make sure the vaccine could be distributed equitably across the country. The update also reminded practices the vaccine has a limited shelf life and said they should only order what they need for a week or two in advance.
The update states: ‘To ensure that vaccine is distributed equitably across the NHS in England for the children’s part of national flu programme, an initial restriction on the number of doses of Fluenz Tetra that can be ordered by each practice per week will be implemented from the start of ordering on Wednesday 24 September.
‘The controls will allow practices to order 50 doses per week (five packs) initially. We will be monitoring the situation on a daily basis and as soon as we are in receipt of sufficient quantities of vaccine we will lift the restriction.’
GPs are this year taking on seasonal flu immunisation of four-year-olds, as well as two- and three-year-olds as they did last year when the routine flu vaccination programme children was first introduced.
However, as all the vaccine stocks will expire by the end of January, PHE is again urging GPs to get as many children immunised before Christmas as possible – which GPC warned could leave practices struggling to fit them in.
A PHE spokesperson told Pulse the restriction was the result of a ‘gradual supply’ of vaccine from the manufacturer, as well as the shorter shelf-life of live-attenuated vaccines.
The spokesperson said: ‘The temporary order restriction is concerned with the gradual supply of the vaccine and the shorter shelf life of the flu nasal spray vaccine Fluenz Tetra which means that it has been agreed with the manufacturer that deliveries will be staggered to ensure that there is sufficient in-date vaccine for patients who present later in the season.’
PHE added there is no restriction on the alternative flu vaccine Fluarix Tetra.
The spokesperson said: ‘There is not an order cap in place for Fluarix Tetra but it should only be ordered for eligible children aged from three years who are contraindicated for Fluenz Tetra. It should not be used for children for whom Fluenz Tetra is suitable and flu vaccine ordered via ImmForm should not be used for patients over the age of 18.’
Pulse July survey
Take our July 2025 survey to potentially win £1.000 worth of tokens

Visit Pulse Reference for details on 140 symptoms, including easily searchable symptoms and categories, offering you a free platform to check symptoms and receive potential diagnoses during consultations.













