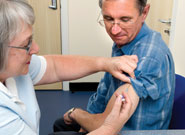
Practices will be unable to order stocks of adult Hepatitis B vaccine ‘until further notice’ after new guidance to ensure sufficient supply for individuals with the highest risk of exposure to the vaccine.
A letter from Public Health England to GPs sent today explains the ordering restriction for each patient type that has been agreed between manufacturers, the BMA’s GP Committee and the Department of Health.
This is in the wake of a global shortage of vaccine stocks which have had an ‘acute impact’ on UK reserves sparking new PHE guidance.
The letter states: ‘It has been agreed with the [GPC] that general practice will not be able to order any adult hepatitis B vaccine stock until further notice.’
Travel to a high risk area is unlikely to be sufficient for vaccination and other cases such as healthcare staff and household contacts of people with hep B should be risk assessed.
The guidance says: ‘A risk assessment should be done on a case by case basis to determine whether vaccination against hepatitis B is indicated but it is unlikely to be available for travel purposes alone.’
GPs are advised not to delay administering paediatric monovalent hepatitis B vaccine for babies born to a hep B positive mother, the highest priority group, with any restrictions on volume of orders able to be overridden.
And the routine childhood immunisation programme starting from late September is also not affected.
The guidance says restrictions are in place ‘to a) prioritise vaccine for post exposure vaccination (particularly infants born to infected mothers); and b) to prevent stock from being exhausted rapidly’.
‘Providers should order only the essential stock for immediate use and should not stockpile. Responsible ordering will help preserve scarce stock for those in greatest need.’
Public awareness materials are under development.
Pulse October survey
Take our July 2025 survey to potentially win £1.000 worth of tokens