UK to switch to non-polio containing pertussis vaccine for pregnant women
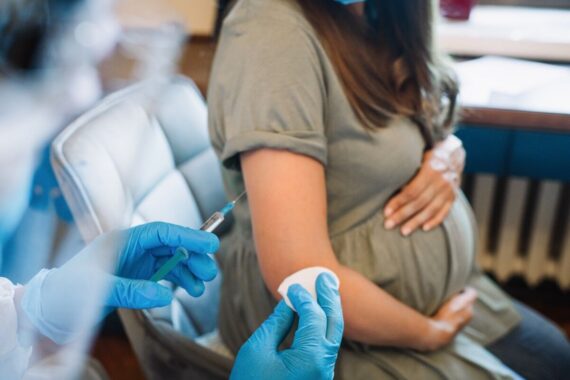
The UK is switching to a different pertussis vaccine for pregnant women following advice from the Joint Committee on Vaccination and Immunisation.
The maternal pertussis vaccination programme will move to using a vaccine that does not contain polio from July, after studies showed that component of the vaccine had a small impact on children’s antibodies later on.
Studies reviewed by the committee at the end of 2022 showed a lower response to polio vaccination at 13 months as well as before and after the preschool booster in children whose mothers had been vaccinated with DtaP-IPV during pregnancy.
Analysis showed antibody levels to polio type 2 were most affected by the maternal vaccination.
The committee noted that despite the difference, all antibody responses were still above the protective threshold and were boosted by the pre-school jab.
To address the ‘potential immunity gap’ caused by the ‘blunting effect’ of the polio in the maternal vaccine, the committee said it would prefer the use of a non-polio vaccine if one could be obtained at a cost-effective price.
The priority should be to ensure that the maternal vaccine programme remains in place because it continues ‘to save lives’, the committee agreed at the time.
Updating the whooping cough vaccination programme advice, UK Health Security Agency officials said they had now secured a supply of ADACEL – a vaccine that contains low dose tetanus, diphtheria and acellular pertussis to use instead.
Any remaining stocks of the low dose dTaP/IPV vaccine (Boostrix-IPV) previously supplied for this programme ‘’should be used for the pre-school booster programme in primary care’, UKHSA said.
The polio-containing vaccine can be used in maternity care settings if it is the only one available until stocks are exhausted and new supplies are received, the advice stated.
It should also be offered if Tdap (ADACEL) is not available or is clinically contraindicated to ensure pregnant women can be vaccinated.
The vaccine is already used in maternal pertussis vaccine programmes in many other European countries, the USA and Australia, with millions of doses administered worldwide, a UKHSA spokesperson said.
Concerns have been raised over falling uptake of whooping cough vaccine in pregnancy after eight babies died in the first four months of this year.
Up to April 2024 there have been 4,793 laboratory confirmed cases of pertussis compared with 858 across the whole of 2023.
In the first few years of the maternal pertussis vaccination programme, coverage was around 70%, but has fallen year on year since 2020.
In 2022/2023 pertussis vaccine coverage was 60.7% and by December 2023, it had fallen to 59.5% in England with some parts of the country seeing uptake below 40%.
Visit Pulse Reference for details on 140 symptoms, including easily searchable symptoms and categories, offering you a free platform to check symptoms and receive potential diagnoses during consultations.
Related Articles
READERS' COMMENTS [1]
Please note, only GPs are permitted to add comments to articles










OK, so we do all (hopefully!) know the difference between dTaP and DTaP; and to whom we should not give the latter, but what is the difference between dTaP and dTap, and which group should not be given the wrong kind of P/p ?