Spironolactone safe and effective in treating acne in women, study finds
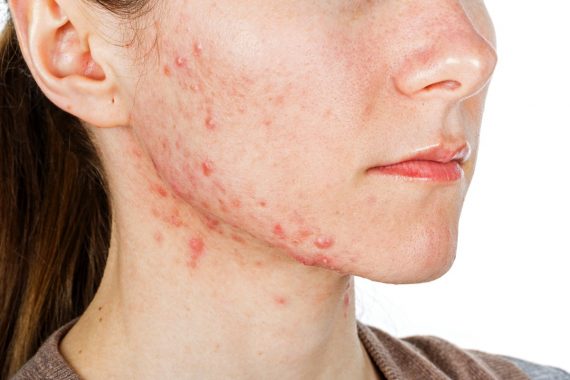
Spironolactone is a cheap, safe and effective for treating women with acne that can also be used in the longer term, a large UK study has shown.
The randomised controlled trial of 410 women over the age of 18 years with mild, moderate and severe acne found a significant difference in both in quality of life and self-reported acne improvement compared with placebo.
Reporting the results in the BMJ, researchers from the University of Southampton said the NIHR-funded trial was the first to provide clinical evidence that spironolactone was effective for the skin condition.
Differences were seen by 12 weeks but were more marked and statistically significant by 24 weeks, they said.
By this time the number needed to treat for participants to report improvement in the condition of their skin was five.
There did not seem to be a difference in effectiveness between women who had PCOS and those who did not, the researchers noted.
It could mean a change in the way the condition is treated including a reduction in the number of antibiotics prescribed, as currently recommended in UK guidelines when topical treatments have not worked, they concluded.
More frequent headaches was the only difference in adverse effects between the two groups who were started on 50 mg spironolactone or matched placebo, increasing to two tablets daily at six weeks if tolerated, which was the case for more than 95% of participants.
Two other trials underway are looking at higher starting doses of the treatment.
Questions relative effectiveness in comparison with other treatments, use in younger age groups and which subgroups benefit would be answered in other studies, the team added.
A linked editorial said the women in the study had already heard about spironolactone as a possible treatment on social media and were keen to try it.
Study author Professor Miriam Santer, a GP and professor of primary care research at the University of Southampton said spironolactone could provide a ‘fantastic alternative’ to oral antibiotics in women where topical treatments had not worked.
‘We hope the publication of these results will mean more GPs and dermatologists feel confident to prescribe spironolactone as a treatment for acne.
‘The drug is already included in treatment guidelines for persistent acne in the US and Europe, and we hope this trial will lead to a change in the UK guidelines.’
Professor Alison Layton from Harrogate and District NHS Foundation Trust and the Skin Research Centre at the University of York, who also worked on the study said dermatologists had been prescribing the drug for severe acne for some time.
‘This is a cheap medication which has been used for decades in the treatment of high blood pressure. The drug also reduces the main hormone that leads to the development of acne.
‘However, previous studies of spironolactone for acne have been very small and there was no definitive proof that it actually worked.’
Professor Andrew Farmer, director of NIHR’s Health Technology Assessment programme, said: ‘The findings from this important trial provide compelling evidence which could help thousands of women affected by persistent acne.
‘The treatment provides a valuable alternative to antibiotics and ensures clinicians can also better avoid the harms that can arise from antimicrobial resistance.’
Visit Pulse Reference for details on 140 symptoms, including easily searchable symptoms and categories, offering you a free platform to check symptoms and receive potential diagnoses during consultations.
Related Articles
READERS' COMMENTS [2]
Please note, only GPs are permitted to add comments to articles














I wonder how many actually go through the detail and explain to patients that it is being used off-license?
It sounds like a useful addition to possibilities, although it is only a 20% response rate (NNT = 5).