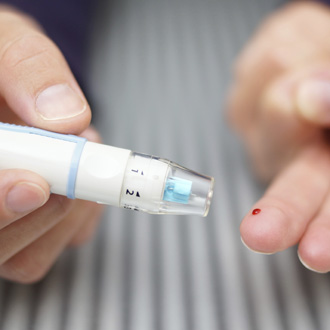
Decisions by CCGs to switch insulin types to save money has led to a number of medicines safety incidents, a pharmacist report has warned.
In a patient safety update report, the National Pharmacy Association said they had received two to three reports of incidents where the local CCG medicine management team has switched patients who were previously stable on Lantus insulin to Abasaglar insulin due to cost saving measures.
The move – in which the patients had not been informed of the change – led to unstable glucose levels.
Switches leading to safety incidents had been done in both adults and children, the update said.
The alert came with a reminder that although both brands contain insulin glargine and are biosimilar of each other, it resulted in the patient becoming hypoglycaemic because they did not know the switch had been made and that they would need to monitor their condition more closely.
In theory moving between the two drugs could require adjustment to dose, the update warned.
Dr Andrew Green, BMA GP committee clinical and prescribing lead, said: ’It is important to realise that biosimilar medications are not direct equivalents in the way that generic medicines are to a brand, as they are biological medicines which are highly similar to another biological medicine already licensed for use, but not identical.’
He said NHS England had issued advice on the use of biosimilar medicines which makes it clear that patients should always be involved in decisions about changes.
’Medicines management teams should be both be aware of this guidance and follow it,’ he said.
Pulse October survey
Take our July 2025 survey to potentially win £1.000 worth of tokens