Rheumatology clinic – pseudogout
Case
A 76 year old lady with insulin-dependent diabetes, osteoarthritis and hypertension presents to her GP with pain and swelling of the right knee that developed overnight. She is unable to put any weight on it, and is accompanied by her daughter. There is no previous history of gout. She has previously seen her GP for bilateral knee pain, which was felt to be related to osteoarthritis. However, the current episode is out of character for her arthritis.
On examination she is apyrexial. The right knee is erythematous, warm, swollen and markedly tender and has a negligible range of movement. Apart from this, she has bilateral hallux valgus and her fingers show changes of nodal osteoarthritis.
Her GP suspects that she has septic arthritis and refers her to A&E urgently.
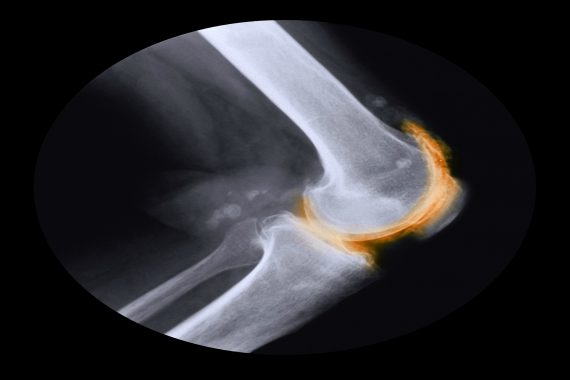
At A&E, she is assessed by the A&E registrar who orders an FBC, kidney function test, LFT, coagulation profile, CRP and ESR. The right knee is radiographed which shows coarse, shaggy articular cartilage calcification arranged in a linear fashion, osteophytes and joint space narrowing.
The A&E registrar reviews the radiograph and thinks that this could be acute calcium pyrophosphate (CPP) crystal arthritis. However, he knows that septic arthritis and acute CPP crystal arthritis can co-exist and performs a knee aspiration after obtaining informed consent.
The synovial fluid is sent to the laboratory for gram stain, examination under polarised light microscopy and culture.
The blood tests demonstrate high CRP (168mg/l), leucocytosis (15,000/mm3) and neutrophilia (80% neutrophils). The liver and kidney function tests are normal and the synovial fluid aspirate shows scattered, weakly positively birefringent crystals, some of which have an intracellular location. The 24-hour synovial fluid culture is sterile and a diagnosis of acute CPP crystal arthritis (previously pseudogout) is made.
The problem
This patient has acute monoarthritis. The commonest cause for this includes acute gout, acute CPP crystal arthritis (especially in the elderly), and seronegative spondyloarthropathy – e.g. psoriatic arthritis or reactive arthritis (in young adults).
Acute CPP crystal arthritis is the most dramatic manifestation of calcium pyrophosphate deposition (CPPD). Other manifestations include asymptomatic chondrocalcinosis, CPPD plus osteoarthritis, and chronic CPP crystal inflammatory arthritis.
Risk factors for CPPD include:
- Increasing age
- Osteoarthritis
- Meniscectomy
- Metabolic conditions such as hyperparathyroidism, haemochromatosis, hypophosphatasia and hypomagnesemia
- Genetics (e.g. polymorphisms in the ANKH and CCAL-I genes)
- Knee malalignment
Septic arthritis and acute gout are the main differential diagnoses. Definitive diagnosis of both these conditions requires aspiration of synovial fluid and prompt examination.
The presentation of patients with hot swollen joints, where joint sepsis is suspected, should be treated as an emergency with prompt referral to on-call disciplines such as rheumatology, orthopaedics and the emergency department. Delayed or insufficient treatment of septic arthritis can lead to permanent joint damage with subsequent morbidity and an estimated case fatality rate of 11%.
Features
Acute CPP crystal arthritis results in abrupt onset (frequently overnight) joint pain, swelling and inability to use the joint. The overlying skin turns red and there may be pitting oedema. Some patients develop fever and systemic illness. The attacks resolve within 10 days to two weeks.
Acute CPP crystal arthritis frequently occurs at the knee and wrist. Elbow, shoulder, ankle and spine (crowned dens syndrome, myelo-radiculopathy) are uncommon manifestations. Tenosynovitis may be present in some patients, especially at the wrist.
Acute CPP crystal arthritis generally presents as monoarthritis. However, it can sometimes present as oligo-arthritis e.g. involvement of both knees or one knee and one wrist.
Most episodes of acute CPP crystal arthritis occur spontaneously with no trigger or precipitant. In some individuals there may be a history of preceding joint injury or recent or concomitant infection elsewhere. Anecdotal evidence suggests that parathyroidectomy, initiation of bisphosphonate treatment, treatment with granulocyte macrophage colony stimulating factor and surgery may precipitate acute CPP crystal arthritis.
Diagnosis
Acute CPP crystal arthritis is not a clinical diagnosis. Although clinical features (elderly patient, swollen knee or wrist) and radiographic examination are suggestive, joint aspiration and prompt examination of the aspirates synovial fluid is of the utmost importance (also to exclude septic arthritis). Routine investigations are listed in table 1. In some patients it may be reasonable to start treatment with intravenous antibiotics for suspected septic arthritis once synovial fluid has been collected, and to stop them when the 24 hour culture results are negative. CPP crystals are difficult to observe under polarised light microscopy and can be missed, leading to a false negative result.
| Table 1: Investigations | ||
|---|---|---|
|
Haematology/biochemistry |
Microbiology |
Imaging |
|
Full blood count |
Blood culture |
X-ray of affected joint(s) – to assess for chondrocalcinosis |
|
Urea & electrolytes, liver function tests |
Synovial fluid gram stain, microscopy and culture1 |
|
|
Inflammatory markers (CRP/ESR) |
Urine dipstick (and culture if indicated) |
|
1Anticoagulant therapy is not an absolute contraindication to joint aspiration.
In the elderly, and in joints with osteoarthritis, CPPD occurs due to age-related changes or as part of the osteoarthritis process. Detailed search for metabolic predisposition can be unrewarding in this population. Expert guidance suggests screening tests should be performed to identify metabolic predisposition to CPPD in those who are younger than 50 years in age, or have widespread polyarticular CPPD. Such individuals should have a detailed family history taken, and screened for presence of haemochromatosis, hyperparathyroidism, hypophosphatasia, and hypomagnesemia. In the elderly screening tests should only be performed to identify hyperparathyroidism as this gets more common with increasing age.
Management
The treatment of acute CPP crystal arthritis includes rest, ice or cold pack application and analgesia. Joint aspiration itself may provide pain relief by reducing intra-articular hypertension. There are no large scale trials of pharmacologic treatment of acute CPP crystal arthritis, and evidence for its treatment is extrapolated from gout studies. Intra-articular corticosteroid injection, once joint sepsis is excluded, is the treatment of choice, especially if only one or two joints are involved.
Long-acting corticosteroids such as methylprednisolone or triamcinolone acetonid (40mg – single intra-articular injection of either agent) with lignocaine are frequently used in clinical practice.
Oral corticosteroids at a dose of 20-30mg/day, for a 7-10 day period may be used, just as in the treatment of acute gout.
Colchicine 0.5mg twice a day for the duration of the episode plus 24 hours may be used. It has a narrow therapeutic index and multiple drug interactions (e.g. with statins, macrolides), so caution should be exercised especially in the elderly and in those with renal impairment. Colchicine can cause diarrhoea, even at this low dose, and may be given once a day in some patients. NSAIDs are likely to be effective in the treatment of acute CPP crystal arthritis, but their use is fraught with risks of gastrointestinal bleeding and renal impairment, and most clinicians avoid using NSAIDs for acute CPP crystal arthritis in the elderly.
If a metabolic predisposition to CPPD is detected, treatment of such diseases is sensible. However, their treatment may not dissolve CPPD and the acute attacks may continue unabated. One small controlled study reported an association between magnesium supplementation and less severe joint pain in those with CPPD. However, magnesium supplementation is poorly tolerated and it is difficult to justify it in those with normal or borderline low magnesium levels. A randomised controlled trial found hydroxychloroquine to be effective in preventing episodes of acute CPP crystal arthritis and may be used. Although anecdotal evidence suggests that methotrexate can reduce the frequency of acute CPP crystal arthritis, a small randomised controlled trial demonstrated no beneficial effects.
The treatment of CPPD plus osteoarthritis is the same as for osteoarthritis alone, with physiotherapy, weight loss, correction of adverse biomechanical factors and analgesia playing key roles.
Dr Abhishek Abhishek is an honorary consultant rheumatologist at the Queen’s Medical Centre, Nottingham and a clinical associate professor of rheumatology at the University of Nottingham.













